Layers and Composition of the Atmosphere
|
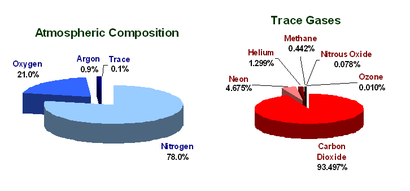
What is the atmosphere?
What are the layers of the atmosphere? |
|
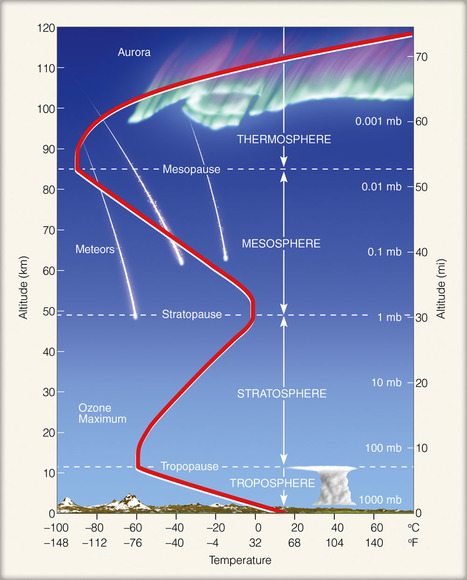
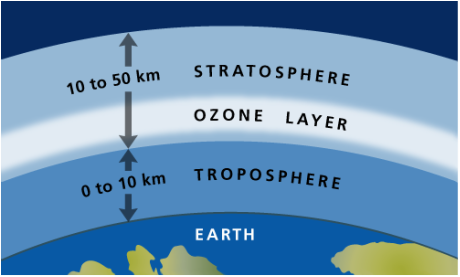
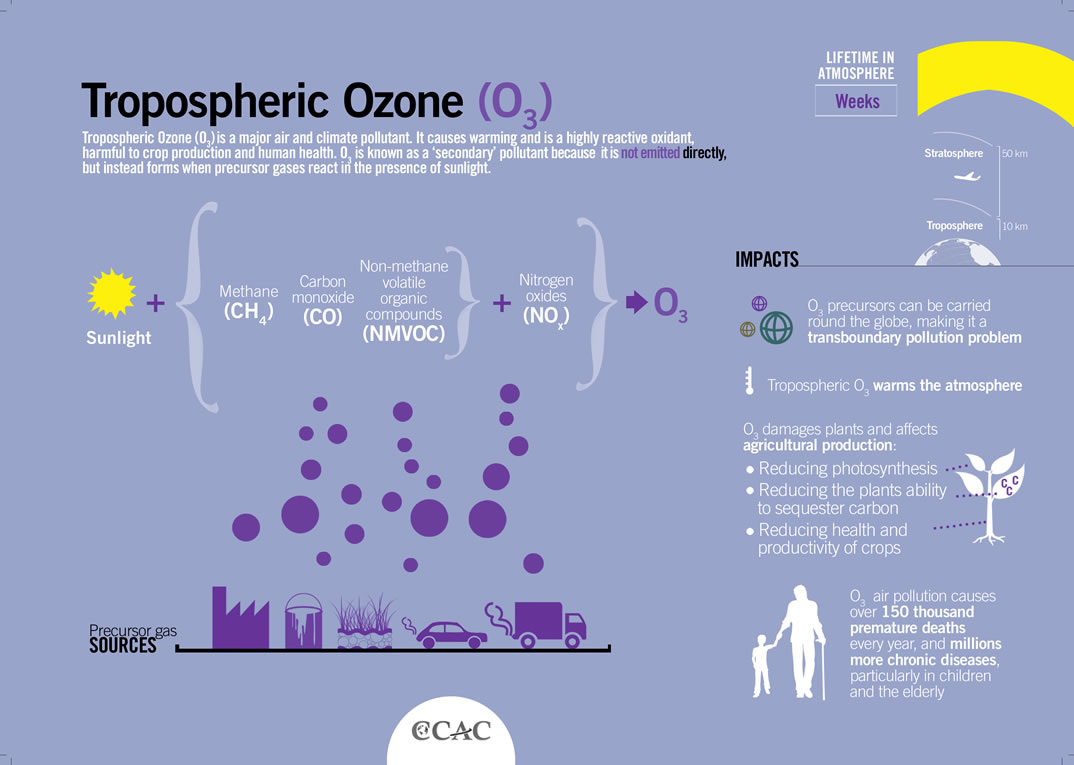
There are four layers of the atmosphere:
Troposphere The lowest layer of the atmosphere is the troposphere. This layer contains most of the clouds and weather, and on average, it extends to a height of 11 km from the Earth's surface, ranging from 8 to 16 km thick. The temperature decreases from 17°C to -51°C as altitude increases because the air becomes less dense at higher altitudes. The tropopause is the boundary between the troposphere and mesosphere and has a stable temperature. Stratosphere The second layer of the atmosphere is the stratosphere. Above the troposphere, it extends from 12 to 50 km above the surface of the Earth. Temperatures increase from -51°C to -15°C as altitude increases due to ozone layer's absorption of the Sun's UV radiation. The stratopause is an area of constant temperature that divides the stratosphere and the mesosphere. Mesosphere The third and coldest layer of the atmosphere is the mesosphere. It is found 50 km to 80 km above Earth's surface, and temperature decreases to -100°C as altitude increases. This layer contains "shooting stars," or meteors that burn when they enter the mesosphere. The mesopause is the boundary between the mesosphere and the thermosphere and has a constant temperature. Thermosphere The thermosphere is the outermost atmospheric layer. Temperature increases to over 1000°C as altitude increases, but because the air is very thin and there are only few air molecules, the air would feel very cold to humans. There are too few air molecules to transfer a significant amount of the heat energy. The atmospheric layer extends from 80 km to over 10,000 km above the Earth's surface, merging with at a certain altitude. The thermosphere can be divided into two sections: the ionosphere and the exosphere. 1. The ionosphere comprises the lower portion of the thermosphere and extends from 80 km to 550 km above the surface of the Earth. The ultraviolet radiation from the Sun is so strong that it ionizes the air molecules, breaking them apart and leaving floating ions and electrons. The high-energy particles from the sun create the colorful lights of the aurorae. 2. The exosphere comprises the second portion of the thermosphere that eventually merges into space. It is found from 550 km to over 10,000 km above the surface of the Earth. It is comprised of mainly hydrogen and helium and at low quantities, as the air is very thin. Satellites are found in this area ("Atmospheric Layers"). |
Montreal Protocol
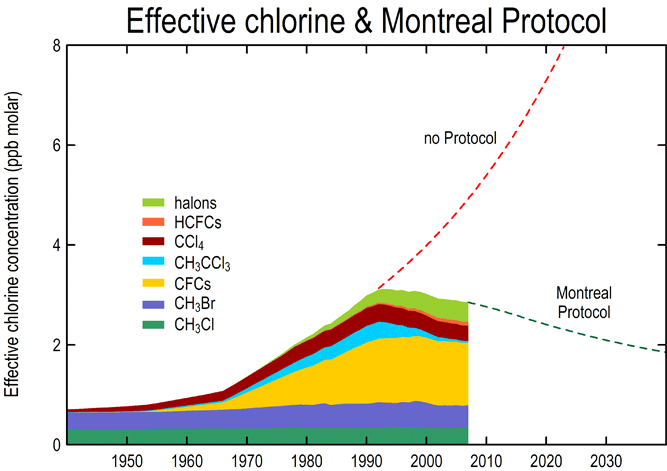
The Montreal Protocol on Substances that Deplete the Ozone Layer is an international treaty to reduce, and ultimately, phase-out the production and emission of ozone depleting chemicals (OCD), such as halons and CFCs, that harm the stratosphere ("The Montreal Protocol on Substances"). Adopted in 1987 in Montreal and implemented in 1989, it has been adjusted 6 times and amended 4 times. Initially, it contained 46 countries but now has almost 200 countries involved [8].
The treaty sets deadlines by which countries must eliminate the production and consumption of certain chemicals. For developed countries, the production of halons ended by 1994, CFCs by 1996, and methyl bromide by 2005, and HCFCs should be eliminated by 2030. For developing countries, CFCs were phased-out by 2010, and methyl bromide and HCFCs are scheduled to be eliminated by 2015 and 2040, respectively ("Montreal Protocol").
The Parties to the Protocol meet each year and make decisions to adopt new techniques and implement adjustments. The treaty has trade sanctions to meet the chemical production limits and to foster multilateral trade and provide economic incentives for non Parties or countries to sign the environmental agreement. A consumption controlling formula was developed to determine the consumption limits and was deemed to be "consumption = production + imports - exports." Moreover, the Annexes of the Protocol list the controlled substances that are banned from importation or exportation of the signed countries. The four Amendments to the Protocol in London, Copenhagen, Montreal, and Beijing were ratified to increase the phase-out rate ("Case Study: Montreal Protocol").
The treaty has been successful, as many scientists find that there has been a substantial decrease in chemicals that harm the ozone over the years that the treaty has been implemented. They believe that the ozone can be recovered beginning in 2020, with a full recovery by 2049 ("Montreal Protocol").
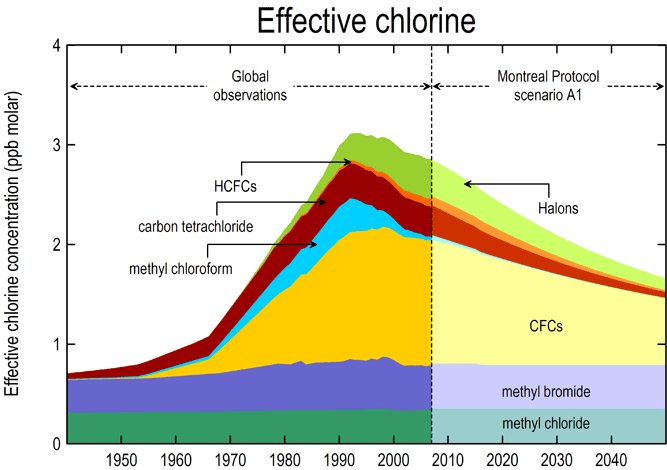
The treaty sets deadlines by which countries must eliminate the production and consumption of certain chemicals. For developed countries, the production of halons ended by 1994, CFCs by 1996, and methyl bromide by 2005, and HCFCs should be eliminated by 2030. For developing countries, CFCs were phased-out by 2010, and methyl bromide and HCFCs are scheduled to be eliminated by 2015 and 2040, respectively ("Montreal Protocol").
The Parties to the Protocol meet each year and make decisions to adopt new techniques and implement adjustments. The treaty has trade sanctions to meet the chemical production limits and to foster multilateral trade and provide economic incentives for non Parties or countries to sign the environmental agreement. A consumption controlling formula was developed to determine the consumption limits and was deemed to be "consumption = production + imports - exports." Moreover, the Annexes of the Protocol list the controlled substances that are banned from importation or exportation of the signed countries. The four Amendments to the Protocol in London, Copenhagen, Montreal, and Beijing were ratified to increase the phase-out rate ("Case Study: Montreal Protocol").
The treaty has been successful, as many scientists find that there has been a substantial decrease in chemicals that harm the ozone over the years that the treaty has been implemented. They believe that the ozone can be recovered beginning in 2020, with a full recovery by 2049 ("Montreal Protocol").
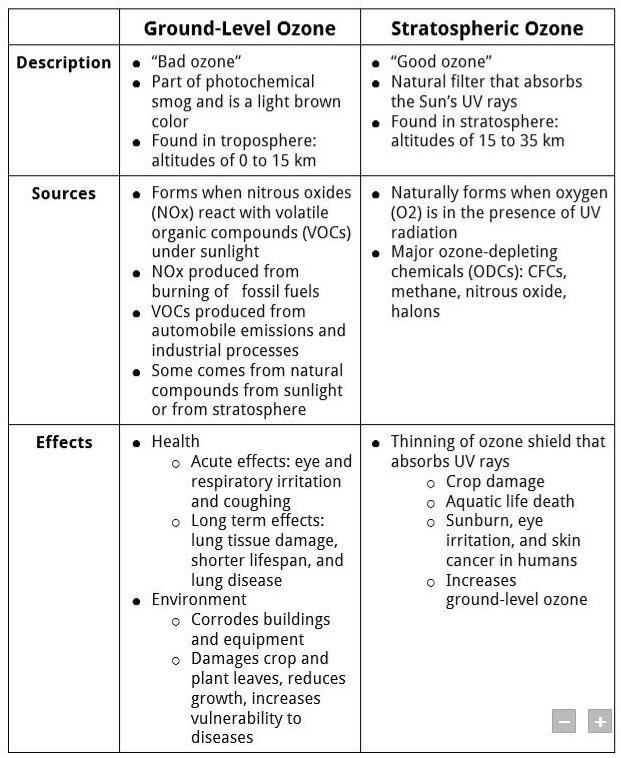
Ground-Level vs. Stratospheric Ozone
What is the difference between ground-level and stratospheric ozone?
Solutions
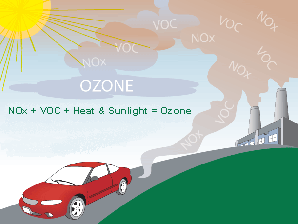
Ground-LevelThe Environmental Protection Agency (EPA) has developed national and local programs and rules to regulate the emission of pollutants that contribute to the "bad," or ground-level, ozone. The EPA has implemented:
You can help reduce pollution by:
|
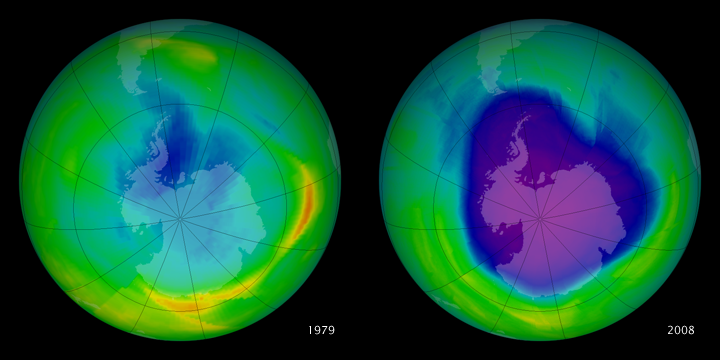
Hole in OzoneWhat is the ozone hole?
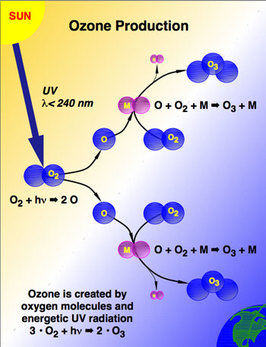
Discovered 30 years ago, the ozone "hole" is an area of ozone depletion over Antarctica. In the winter, winds and ice crystals on clouds release CFCs through chemical reactions, which deteriorate the ozone layer. In the ozone hole, the concentration of ozone decreases to less than 220 Dobson units. What causes ozone depletion?
Artificially and human produced chemicals such as halons and CFCs are responsible for the thinning of the ozone layer. These ozone-depleting chemicals (ODCs) are dispersed through the atmosphere and react with UV rays to produce chlorine and bromide atoms. These atoms destroy the ozone, each atom destroying over 100,000 ozone molecules ("The Process of Ozone Depletion"). Weather conditions can also affect the size of the ozone hole, as it reached its largest size in 2006, extending over 11 million miles, but has significantly shrunken in recent years. As the ozone layer, which blocks UV rays, thins, the Earth is more exposed to harmful UV radiation that can cause reproductive and growth issues in plants an animals, as well as skin cancer in humans ("The Ozone Hole"). What is being done? The Montreal Protocol of 1987 was a global effort to reduce the production and consumption of CFCs and other ozone-depleting chemicals (ODCs), and the treaty was signed by almost 200 countries. It has been successful in significantly reducing and banning all production of CFCs, and scientists predict that it may restore the ozone layer to its original size by 2049. Moreover, the United States passed the Clean Air Act of 1990, which required the NOAA and NASA to continuously oversee the ozone hole (Harankhedkar, 2011). What can you do?
|
Case Study
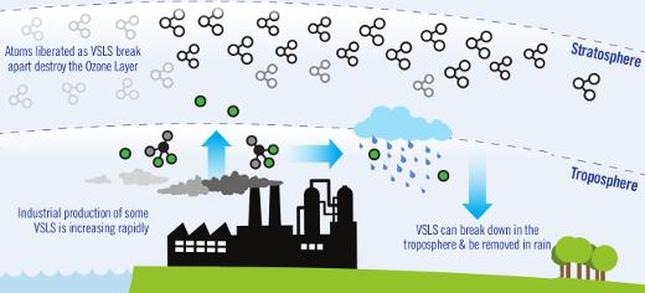
Scientists at the University of Leeds have discovered that VSLS (Very Short-Lived Substances) are increasing in concentration in the atmosphere and may be contributing to ozone depletion. Dichloromethane is the one causing the most concern now as previously, the concentrations were not high enough to cause noticeable destruction. VSLS are extremely dangerous because they are short-lived; they do not have the time to float up through the atmosphere before destroying ozone. VSLS destroy atmospheric ozone that is in the lower part of the stratospheric zone, which poses much higher risk to climate than does ozone destroyed higher up. (University of Leeds, 2015)
http://www.sciencedaily.com/releases/2015/02/150216130241.htm
http://www.sciencedaily.com/releases/2015/02/150216130241.htm